doi:10.1016/j.provac.2012.04.015Ĭoncentrating the product of interest in situations where there is a large impurity level in the feed stream such as those found in early stage monoclonal antibody purification where typically resin based affinity or cation exchange chromatography is used. Selection and/or peer-review under responsibility of the 5th vaccine conference organizing committee. Nakry Sisowath is presently at 200 Prospect Street, Waltham, MA 02454-9141 U.S.A.įranklin Chung is presently at Novartis 350 Massachusetts Avenue # 1 Cambridge, MA 02139-4182 USA.ġ877-282X © 2012 Published by Elsevier Ltd. Ltd., 35 Tuas South Ave 6, Singapore 637377, Singapore. E-mail address: Ramaswamy is presently at Lonza Biologies Tuas Pte. As a result this method may require significant amount of buffers, post processing of product and longer processing and turnaround times. The molecule of interest is then recovered from the stationary phase by changing fluid parameters such as conductivity, pH, polarity and chaotropicity.
In this approach the molecule of interest is bound to a stationary phase and the undesired impurities are allowed to flow through the column along with the mobile phase. Traditionally liquid chromatography has been used in bind and elute mode which is at bare minimum a three step batch operation.

Keywords:influenza virus adsorber beads flow-through purification host cell protein DNA.
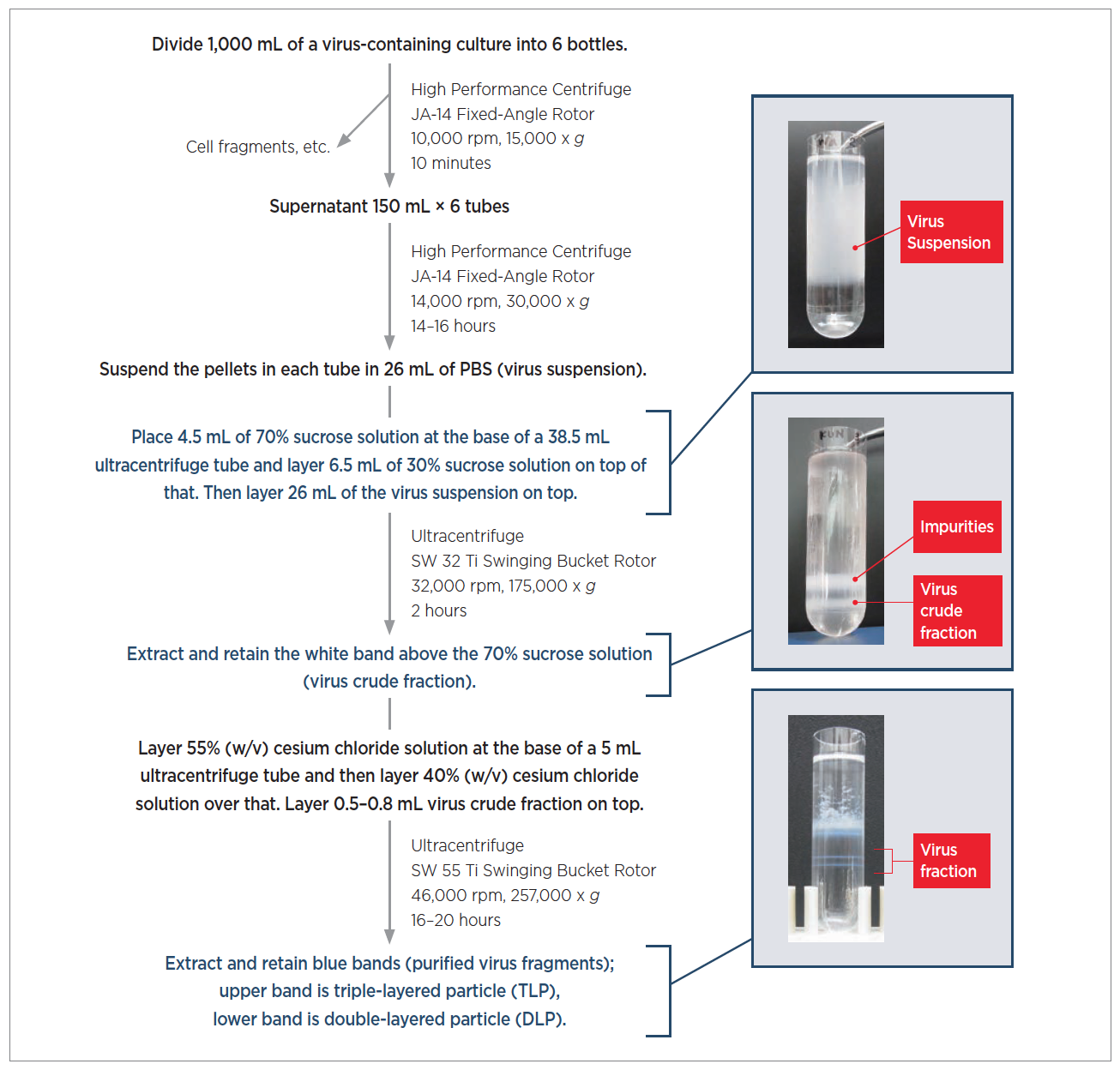
Selection and/or peer-review under re sponsibility of the 5th vac cine conference organizing committee. The column cleared > 80% of the HCP and the membrane adsorber reduced whole hcDNA levels to <10 ng. Virus recoveries of >70-80% and 100% were achieved for the column and membrane approaches respectively. The techniques were evaluated using cell culture grown influenza virus and bacteriophage feed streams. This enabled selective binding of host cell DNA (hcDNA) to the membrane and complete recovery of virus in the flow-through mode. For the second step a chromatographic separations strategy using a primary amine membrane adsorber and multivalent ions in the mobile phase was developed. A resin column with reduced surface area was developed for the first step to remove bulk of the host cell protein (HCP) from a viral feed stream while allowing most of the virus to flow-through. Ganesh Iyer*, Senthilkumar Ramaswamy, Kwok-Shun Cheng, Nakry Sisowath, Ushma Mehta, Anne Leahy, Damon Asher, Franklin ChungĮMD Millipore, 80 Ashby Road, Bedford MA, 01730, USAĪ two step flow-through chromatography process is proposed as an universal approach to purify viruses. Procedía in Vaccinology 6 (2012) 106 - 112ĥth Vaccine and ISV Annual Global Congressįlow-Through Purification of Viruses- A Novel Approach to Available online at Procedía in Vaccinology

Abstract of research paper on Chemical sciences, author of scientific article - Ganesh Iyer, Senthilkumar Ramaswamy, Kwok-Shun Cheng, Nakry Sisowath, Ushma Mehta, et al.Ī two step flow-through chromatography process is proposed as an universal approach to purify viruses.
